The «peptide rush» of nowadays, which is observed in the pharmaceutical market and in the plans of research work of scientific laboratories, is largely due to the fundamental scientific achievements of recent years in understanding the physiology, pharmacology and safety of substances of peptide nature, technological advances in the creation of pure synthetic analogues of natural peptides. By now, about 2000 oligo — and polypeptides have been obtained, including real and potential medicine, and their number is only expanding. An important discovery of recent years has been the data that dipeptides, tripeptides, some oligopeptides can be absorbed from the intestine without splitting to individual amino acids and enter various organs and tissues, in particular the brain, producing a distinct therapeutic effect. Nowadays, on the basis of dipeptides, the molecules of which contain two amino acid residues, effective medicines of different pharmacological profile of action have been obtained: psychoactive, actoprotective, immunotropic medicines, remedy of metabolic therapy, which have been clinically tested and have found a wide practical application. As it has been shown by preclinical and clinical studies, therapeutic doses of medicine with peptide nature, as a rule, in 2 or 3 times lower than the therapeutic doses of profile non-peptide analogues. Taking into account the practical absence of allergic reactions to parenteral and enteral introduction of short peptides, relatively small total amounts of peptide medicines recommended for course use, as well as the technical capabilities of creating exact copies of natural oligopeptides, the latter can be considered as a promising class of biologically active substances with a large spectrum of pharmacological potential.
The needs of pharmacological correction of the human activity in extreme conditions of hard work as well as sports of the highest achievements have become the real basis for the search and creation some tools of development and support of the organism’s adaptive potential, and means to optimize the use of organism’s reserve capabilities.
The present article is devoted to the features of the properties and mechanisms of action of dipeptide IPH AG a relatively new means of peptide nature, and in the authors’ opinion, according to the proved based spectrum of activity, can be attributed to the number of means which are able to increase the sustainability and resistance of the body to a wide range of extreme effects.
Peptide bioregulator is a white crystalline molecule consisting of two amino acid residues: alpha-L-amino acids alanin and alpha-L-amino acids glutamine, covalently bound by a peptide bond that is similar to the current bond in proteins (Figure 1). Both amino acids are natural essential physiological substances, use as precursors of protein synthesis, and also are included in the 20 standard amino acids that play an important role in the synthesis of many natural proteins of the body. Both are important for global nitrogen metabolism in the body, gluconeogenesis, energy supply, plastic metabolism and many other physiological processes.

Dipeptide implements its action in different ways of introduction into the body and, depending on the way of entry, is carried out either the action of molecules AG, and/or dissociated molecules of alanine and glutamine. By oral or enteral introduction, alanilglutamine acts locally in the gastrointestinal tract, helping to protect the integrity of the intestinal mucosa, and maintaining the barrier functions of the intestine. This reduces the possibility of bacterial translocation, the risk of infection, infection-induced inflammatory damage and infection-related symptoms such as diarrhea, dehydration, malabsorption and electrolyte imbalance. There is the fact of the ability of the actual dipeptide at different ways of introduction to support the integrative function of the intestine, accelerating the absorption of water and electrolytes from the intestine, a number of macro-and micronutrients, thereby providing a rehydrating effect and increasing the subsequent absorption of proteins and fats. The chronological analysis of development by introduction internally allowed to say about a relative dividing dipeptide effects on urgent — developing within an hour and associated mainly with rehydration and improvement of the functions of excitable tissues, and delayed- developing in hours and days after introduction into the body, and expressing by a stable anabolic and anti-catabolic effects, increased immunity, increased glycogen reserves in muscles, etc. Besides this, it is noticed that Ala-Gin is an effective supplier of alanine and glutamine[1,2,3] and thus mediates the differential action of these amino acids corresponding to the current needs of the body. Protein synthesis is carried out in the body constantly, and interchangeable amino acid α-L-alanine (А- C3H7NO2) is included in many proteins, especially muscle, participating almost continuously in the turnover throughout life. The ability of alanine to easily convert into glucose causes the functioning of the glucose-alanine cycle — one of the main ways of gluconeogenesis in the liver.
Alanine is involved in the formation of muscle proteins, carnosine dipeptides, coenzyme A, the enzyme alanine aminotransferase, Pantothenic acid (vitamin B5). All this determines the basic physiological tasks of the present amino acid: maintaining a balance of nitrogen and a constant concentration of glucose in the blood. This amino acid is one of the most important sources of energy. In the human body, alanine is formed in muscle tissue from lactic acid. With intense physical activity, the lack of alanine stimulates catabolic processes in muscle tissues. Alanine can be formed from other nitrogenous substances or during the decay of carnosine.
L-Glutamine (G- C5H10N2O3 amid monoamino dicarboxylic glutamic acid) is relatively an essential amino acid. The concentration in the blood is 500-900 µmol/l, which is higher than the concentration of any other amino acid and this is a popular amino acid for dietary supplements, it is one of the most well-known candidates for the correction of the performance of persons subjected to increased physical activity, to optimize the physiological functions of athletes, as well as a known means of metabolic therapy in the clinic. Glutamine, being the most abundant free amino acid in the human body, along with glutamic acid, is a precursor to peptides, proteins, neurotransmitters, nitrogenous bases and is used as an energy source by various organs such as the intestine. This amino acid is involved in the realization of many functions, such as maintenance of cell proliferation (cell division), regulation of immunity and cellular activity, supporting of acid-base balance and regulation of gene expression [4,5,6]. In addition to its role as a component of protein, it is important as a global nitrogen transporter, carries out the transamination of amino acids and participates in the formation of new amino acids. Glutamine deficiency reduces the proliferation of a number of cells and stimulates apoptosis (programmed cell death). The introduction of glutamine has a positive effect on glucose metabolism, including insulin resistance. It is proved that the deficiency of L-Glutamine leads to increased fatigue, reduced muscle strength, endurance and attention, increased reaction time and a number of other adverse events that worsen performance in general and sports performance in particular. However, various studies in recent years have shown that the widespread use of L-Glutamine as a separate product is hampered by the peculiarities of its physical and chemical properties, such as: weak solubility and partial decomposition in an aqueous medium with the release of harmful ammonia gas, low thermal stability, pronounced dependence on the pH of the solvent, instability in an acidic medium.
The scientific literature presents a large factual material on the positive effects of L- Glutamine in the dosage range of 0.2-0.4 g/kg/day in athletes and persons engaged in regular exercise [7.8, 9.10, etc.].
At the same time, a number of publications reported that there is no evidence of the effectiveness of L-Glutamine and hypertension in the training process, especially in persons with highly adaptive nutritional status [10, 11].
It can be thought that these differences are primarily due to the large variability of the studied groups of individuals, differences in methodological approaches, the incompatibility of the recorded parameters, as well as the instability of L-Glutamine in the acidic environment of the stomach and other factors. However, glutamine is used in clinical practice as an oral, parenteral or enteral Supplement in the form of a separate amino acid or in the form of glutamine-containing dipeptides, which, as it turned out, have a number of advantages compared to it.
In particular, IPH AG and other dipeptides exhibit greater physical and chemical resistance, provide better delivery and in greater quantities to the organs, tissues and intercellular space of glutamine and alanine. Thus, the water solubility at room temperature and stability in a solution substantially higher than the initial free amino acids alanine and glutamine, and higher than that widely used in sports and clinical pharmacology dipeptide. The ranked number of substances under consideration, in order of decreasing their solubility in water.
Characteristics of physical and chemical properties of these substances are extremely important for the assessment of their pharmacological properties and should be taken into account for the prediction of digestibility in a particular way of introduction into the body, evaluation of biological distribution in the body, calculation of dosages of ingredients and for the correct comparison of physiological efficiency. Thus, the water solubility of L-Alanyl-L-Glutamine is 15 times higher, L-Glycyl-L-Glutamine is about 4 times higher than L-Glutamine. Along with this, instability of L-Glutamine in aqueous solutions, low resistance in the acidic environment of the stomach and relatively slow and incomplete absorption in the intestine were revealed (Table 1).
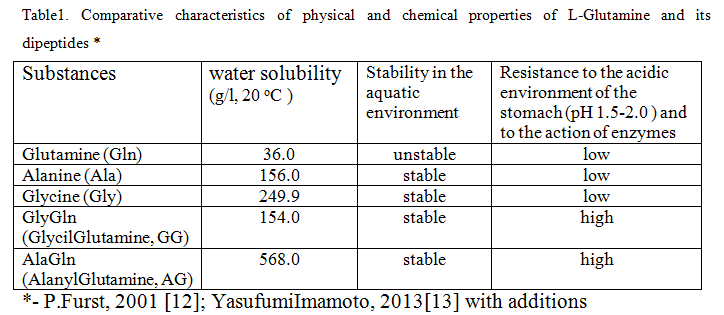
It has been proved that within the first hour at normal temperature of the body is destroyed about 50% of ingested L-Glutamine, whereas dipeptide is stable for at least four hours, creating an environment conducive to the full absorption in the intestine. In addition, the thermostability of dipeptide is much higher than that of glutamine, which decomposes upon heating with the formation of toxic ammonia.
These and other features, which are discussed below, are the advantages of dipeptide as a pharmacological agent. So, due to the stability of depeptide in aqueous solutions, along with dry and powdery forms (usually dry crystalline powders or more often encapsulated powders, under the name L-Glutamine) medicines have been created in the form of a concentrated 20% aqueous solution for parietal nutrition, which is intended for drip intravenous (in) infusion into the Central veins after compatible infusion solution. The dose depends on the severity of the patient’s hypercatabolic state and amino acid requirements.
The recommended maximum daily dose for the medicine is 2.5 ml/kg, which is equivalent to 0.5 g/kg of pure dipeptide, i.e. 3,5 g/day for a person weighing 70 kg. After the introduction of dipeptide is rapidly hydrolyzed in plasma to form alanine and glutamine. The dipeptide life time(T1/2) is 2.4-3.8 minutes. The introduction of dipeptide regulates nitrogen balance and protein metabolism, maintains the intracellular pool of glutamine, correct catabolic response, improves immune function, reduces the frequency of infectious complications, restores the function of the intestine [14].
Infusion of dipeptide leads to a rapid increase in the concentration of glutamine and alanine in organs and tissues, primarily in muscles and liver, during the entire period of infusion and only trace amounts of dipeptide can be detected in plasma.
Comparative study of pharmacokinetics of oral introduction of dipeptide and glutamine demonstrated that in L-Glutamine equivalent doses — dipeptide (89 mg/kg) to a greater extent than the free form of L-Glutamine (60 mg/kg), provides a long and significant increase in the concentration of L-Glutamine in blood plasma. The initial concentration of L-Glutamine is 475 ±108 µmol/l. After 30 minutes of L-Glutamine administration, the amino acid concentration increases by a maximum of 179 ± 61 µmol/l with a return to the initial values after 2 hours. The mean area under the concentration curve (AUC) between 0 and 4 hours was 127 ± 61 µmol∙h∙l-1.
After the introduction of dipeptide, the peak of increasing the concentration of L-Glutamine in plasma was +284 ± 84 µmol/l (to the base values), which is 59% higher than after the introduction of L-Glutamine (P < 0.05). The duration of the increase in the concentration of L-Glutamine was also longer in the case of dipeptide, and the average value of AUC was 284 ± 154 µmol∙h∙l-1, which is higher in two times more than in the case of L-Glutamine (P < 0.05). In a clinical study, it was found that the introduction of dipeptide not only exceeds the free form of L-Glutamine in the rate of absorption in the intestine by more than 2 times, but also retains this ability in chronic inflammation and reduced gastric secretion. Such features of properties indicate the prospects of practical application of dipeptide in clinical and sports medicine [14,16].
In comparative terms, dipeptide is more stable in the gastrointestinal tract, better absorbed in the digestive tract and absorbed by the liver orally than free L-glutamine, often used intravenously in clinical nutrition [17].
Moreover, with oral introduction, dipeptide is more effective for increasing glutamine levels in muscles than the equivalent amount of glutamine. Perhaps this is due to the fact that after intravenous hypertension dipeptide quickly hydrolyzed into molecules A and G, and after oral introduction the molecules of dipeptide work in place in the small intestine of the digestive tract, where the absorption occurs, then a small part of the molecules of dipeptide and free amino acids is transported with lymph in a large circle of blood circulation, and the main part of portal blood is transported to the liver. Further, amino acids, it is possibly that short peptides and proteins synthesized in the liver are transported to other tissues.
Intensive physical activity is a powerful physiological stress, which during the period of the stress factor limits and even turns off the ability of the intestine to fully absorb proteins, fats and carbohydrates, reduces their maximum transportable volume. Chronic intensive physical activity often leads to a number of gastrointestinal disorders, especially in those sports that require increased resistance [18].
Therefore, dipeptide IPH AG is recommended to be used both before prolonged exercise to improve the absorption of electrolytes and increase endurance, and after intense loads to restore the absorption capacity of the intestine. The method of the dipeptide’s use can be similar to the method of use of glutamine.
It has to noted that both alanine and glutamine are important essential substances. Glutamine is the most common amino acid in free form in human cells and intercellular space. Glutamine is synthesized in the human body in a sufficient amount for a normal lifestyle, however, not being an essential amino acid, because of its Central role in global nitrogen metabolism and participation in many regulatory and metabolic processes is considered conditionally indispensable [19, 20].
In addition to its role as the most common protein component and importance in amino acid transamination, glutamine is an important component of various metabolic processes. Glutamine is metabolized in almost all organs and tissues and can be classified as a true regulator of amino acid balance. In extracellular fluid, glutamine is about 25%, and in skeletal muscle more than 60% of the total pool of free amino acids. The transmembrane concentration gradient in muscles is about 34:1 (intracellular/extracellular fluid). The concentration of free glutamine varies greatly in different organs and tissues. It is important that the blood plasma contains only a very small part of the free glutamine in the body and the concentration of this amino acid in the plasma does not directly depend on the intracellular concentration, so, the concentration of glutamine in the plasma can not serve as a marker of glutamine in the body [26]. The total content of glutamine in the body is mainly determined by the proportion of this amino acid in the protein: 4.3±0.6 g per 100 g of muscle protein. Muscles are the main endogenous source of glutamine. Taking into account the fact that muscles make up 40% of body weight, it is believed that the total content of glutamine in muscles in a man of weight 70 kg is approximately 240-280 g [2,12].
At extreme physical pressure or critical pathological conditions, free glutamine is depleted very quickly, the body compensates for the level of free glutamine primarily due to the breakdown of muscle proteins and increased glutamine synthesis. The reason for the development of glutamine deficiency – a large number of metabolic reactions and functions that directly or indirectly depend on glutamine, as well as high and rapidly increasing demand for it in rapidly growing cells.
Moreover, glutamine serves as an inter-organ nitrogen Transporter in the body. Approximately 1/3 of all nitrogen is transported in the blood in the form of glutamine [21]. Thus, the endogenous reserves of glutamine and the possibility of its synthesis are such that in conditions of exhausting loads or aggressive stress relatively quickly develops relative deficiency of glutamine.
Most of the nitrogen consumed by muscles is used in muscle cells to synthesize glutamine, which is a non-toxic carrier of ammonium from peripheral tissues to internal organs. Glutamine is the main substrate for the synthesis of urea in liver ammoniagenesis in the kidneys. In mitochondria with glutaminase glutamine can turn into glutamate with the formation of ammonium. In general, Glutamine, as an interorgan nitrogen carrier, is important in the excretion of protein metabolism products (urea, creatinine, uric acid, purine bases, etc.) and maintenance of acid-base homeostasis. In the kidneys with the renal isoenzyme glutaminase glutamine is used for ammoniogenesis with the consumption of Н+ ions.
Additionally, it has to note that both amino acids are vital not only for the exchange of nitrogen between tissues, but also for the production and / or storage of glucose. Moreover, the functions of alanine and glutamine are realized not only in parallel, but they are often interrelated and synergistic: alanine is the most important amino acid involved in the transfer of nitrogen from the muscles to the liver. Once there, the carbon skeleton of the amino acid can be converted to glucose as part of the glucose-alanine cycle [22].
Both amino acids also improve hydration in synergistic ways. Apparently, Alanine affects hydration and cell volume by increasing intracellular potassium concentrations, thereby drawing water into the cell. Glutamine also affects the volume of cells, helps to maintain the overall acid-alkaline balance of the body, due to ammonia, which is cleaved from the amino acid and delivered to the kidneys, affects the transport of water. In response to heavy physical exertion or other forms of physiological stress, both amino acids are released from the muscles in significant concentrations. If the concentrations of these amino acids in the muscles are not restored, then resistance to physical activity, inhibition of recovery, decrease in the functions of the immune system, muscle growth, General hypofunction may decrease. Healthy people of weight about 70 kg have from 70 to 80 g of relatively free glutamine distributed throughout the body. In quantitative terms, skeletal muscles are the most significant pool of glutamine [23]. Using isotopic and pharmacokinetic methods, it was found that the endogenous production of glutamine ranges from 40 to 80 g / day [24-26]. In plasma obtained from blood samples, the concentration of glutamine varies from approximately 500 to 800 µm / l (values recorded after 12 hours on an empty stomach), which is about 20% of the total pool of free amino acids in the blood [27]. In tissues such as the liver and skeletal muscles, the concentration of glutamine is even higher than in plasma, which is about 40-60% of the total amino acid pool. As in plasma, and in tissues especially muscular, glutamine concentration in 10-100 times higher than any other. The total reserve of glutamine muscle reserve is relatively small (in a person with a mass of 70 kg about 240 g), the stress-induced decay of 1 kg of muscle tissue provides only 9 g of glutamine, so the catabolic release of glutamine is limited and insufficient for prolonged physical pressure of depleting action [23].
Thus, skeletal muscles play a critical role in glutamine metabolism, as muscles are one of the most common tissues in the human body [28]. Intramuscular glutamine content corresponds to 50-60% of the total free amino acids in the tissues of skeletal muscles. Approximately 80% of glutamine in the body is found in skeletal muscles, and this concentration is 30 times higher than in human plasma [29,30]. The concentration of free amino acids in muscle tissue depends on the type of muscle fiber. Studies on rat skeletal muscle showed that glutamine concentrations were three times higher in slow muscle fibers (type 1 fibers) than in fast muscle fibers (type 2 fibers). High concentration of glutamine in the slow twitch muscle fibers due to the high activity of the enzyme GS and the availability of ATP for the synthesis of glutamine [31].
Glutamine is an important source of carbon and nitrogen for various substrates in the body. G is used directly for protein synthesis and serves as a precursor for the synthesis of other amino acids. Glutamine is a donator of nitrogen for the synthesis of aminosaccharides, purines and pyrimidines used for the synthesis of nitrogenous bases that are part of deoxyribonucleic (DNA) and ribonucleic (RNA) acids necessary for cell proliferation and protein synthesis [32].
In case of critical stress, there is a functional redistribution of the supply of organs with plastic and energy resources, the states of hypercatabolism and hypermetabolism develop, the balance between the production and consumption of glutamine is disturbed. The small intestine is the main organ that consumes glutamine. Under stress, the use of glutamine by the gut increases, which increases its deficiency.
Today it is proved that glutamine is an absolutely necessary substrate to maintain the structure and function of the intestine (33, 34), especially in conditions when there is damage to the intestinal mucosa, deterioration of barrier function and, consequently, an increase in the degree of translocation of bacteria and toxins into the bloodstream [33-35]. If hyper catabolism is not corrected, the risk of multi-organ failure increases. It is suggested [34] that increased consumption of glutamine under stress can save glucose for organs that obligate use it for energy: the brain, red blood cells, bone marrow and granulation tissue. It is shown that rapidly dividing cells, including cells of the intestinal mucosa, pancreas, pulmonary alveoli and cells of the immune system, use glutamine for energy and plastic needs. Glutamine is the main source of energy for cells of the gastrointestinal tract (enterocytes, colonocytes) [33].
The physiological state of the small intestine is highly important for the absorption of substances primarily of protein nature, because it is in this part of the gastrointestinal tract that acid degradation of polypeptide molecules is completed, their enzymatic hydration and absorption occurs: oligonucleotides, short peptides (tripeptides, Alanil-Glutamine and other dipeptides), as well as amino acids (Figure 1). An important role in the breakdown and absorption of peptides play borderline enterocytes, located on the villi of the intestinal epithelium. These cells are involved in almost all stages of digestion, including parietal and membrane.
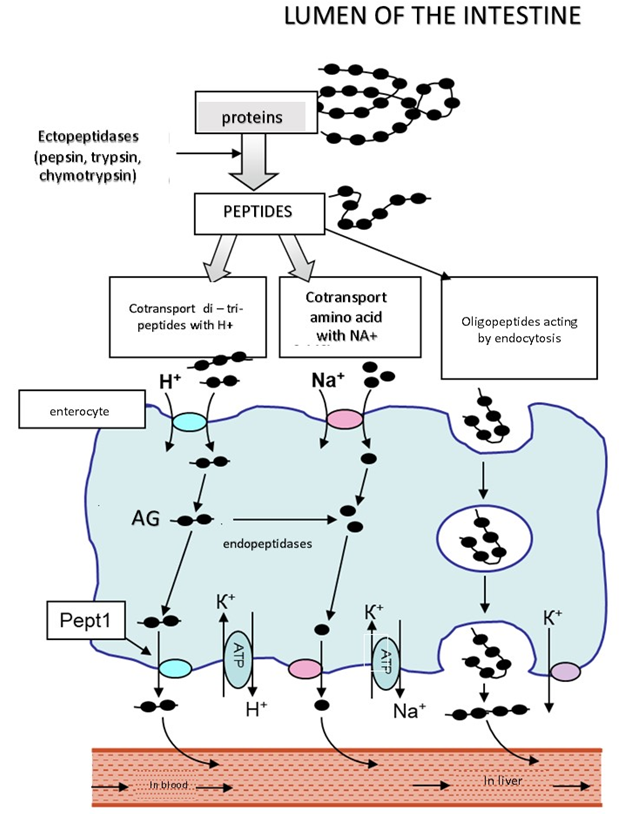
Fugure 1 Scheme of transport of amino acids and substances of peptide nature (oligopetides, di- , tripeptides) through the cells of the small intestine (enterocytes) in the portal vein of the liver and lymphatic bed. Transport of dipeptide is a proton (H+)-bound cotransporter РерТ1.
It is now established that polypeptides are broken down under acid and enzymatic action in the lumen of the intestine to oligopeptides, peptides consisting of two or three amino acids to free amino acids. Forming di-and tripeptides accumulate in significant amounts in the cytosol of cells(enterocytes) of the intestinal mucosa. Some of these peptides are hydrolyzed to free amino acids, but some di-and tripeptides are able to persist in cytosol hydrolysis and be transported intact through the basolateral membrane, joining the transport protein Pept1 – proton (H+)-bound cotransporter (36). Getting mainly into the venous blood of the portal bloodstream and further into the liver and lymph. Thus, with oral or enteral introduction, dipeptide acts locally in the gastrointestinal tract both to protect the integrity of the intestinal mucosa and to maintain the barrier functions of the intestine. These effectives are the basis of the positive influence of dipeptide –maintaining and/or restoring integrative function of the intestine, the normalization of the absorption of nutrients, prevention and inhibition of the development of wasting syndrome (increase of lean body mass – LBM), a correction of the balance of the metabolism by increasing the anabolic and inhibition of catabolic processes in muscle, lung and nerve tissues.
In this regard, among the «urgent effects» of dipeptide, the actions associated with the acceleration of the processes of rehydration of the body at high prolonged physical pressure are justified that there is a preservation of efficiency for a longer time and with high efficiency. Also dipeptide provides a significant ergogenic advantage by increasing the time of endurance under moderate hypohydration stress (37-40).At the same time, in a number of works it is noted that dipeptide not only exceeds the free form of L-glutamine in terms of the rate of absorption in the intestine more than twice, but also retains this ability in systemic chronic inflammation and low secretion of the stomach. In general, such features of the action of hypertension can be of direct practical importance for the use of dipeptide in clinical and sports medicine.
A number of studies have found that rapidly dividing cells, including cells of the intestinal mucosa, pancreas, pulmonary alveoli and cells of the immune system, use glutamine for energy and plastic needs. And for cells of the gastrointestinal tract (enterocytes, colonocytes) glutamine is the main source of energy [33, 41].
During the intracellular oxidation of glutamine is formed ATP which is the most significant molecular form of conservation and use of chemical energy in cells, the total amount of stored energy depends on the availability of glutamine and the degree of its oxidation. In stress environment, this is mainly determined by the level of glutamine deficiency, the availability of glucose as an alternative source of energy in some tissues and the phase of the cell life cycle. For example, lymphocytes use glutamine for energy to a greater extent after mitogenic stimulation (41). Under physiological conditions, glutamine oxidation gives about 1/3 of the energy in rapidly dividing cells [42], with strong stress and pathological reactions, glutamine oxidation can increase.
The functioning of the immune system is also envied by the availability of glutamine. Stress, causing glutamine deficiency, disrupts the immune system. The consumption of glutamine by cells of the immune system is increased by 10 times compared to other cells [43, 44,45].
Glutamine is an indispensable substrate for the normal functioning of humoral and cellular immunity. Thus, in vitro studies have shown that the lack of glutamine in the tissue culture environment severely limits the ability of lymphocytes to respond to mitogenic stimulation [46]. In addition, some inflammatory mediators (interleukins, etc.) and glucocorticoids increase the activity of lymphocyte glutaminase, including in mesenteric lymph nodes, which leads to increased utilization of glutamine.
Consequently, the use of a unique intestinal Transporter exogenously in the form of a molecule of the dipeptide can cover the possible relative scarcity and more efficiently to increase the reserve amount of glutamine in the bloodstream, extracellular space in skeletal muscle and cells in comparison with conventional free glutamine. With the introduction of dipeptide is supported by a higher level of concentration in the glutamine compartments: blood plasma, lymph, intercellular space and skeletal muscle, compared with the introduction of a free glutamine. This feature of the biodistribution is very important because up to 65% enter oral by free glutamine can be destroyed before it reaches the muscles. It can be assumed that in a healthy body there is a multiparameter dynamic homeostasis of glutamine, it is balanced and configured in such a way that an unstable reserve amount of glutamine is maintained. Current needs are met by the reserve and release of glutamine from the muscles and lungs through the breakdown of its own proteins, and replenishment is due to external revenues and increased synthesis of glutamine denovo. Glutamine is actively spent primarily on maintaining a normal structure and the functions of the intestinal mucosa, hepatic ammoniogenesis, proliferation of lymphocytes, other cells of the immune system, muscle function. A decrease in intramuscular glutamine concentration causes a significant increase in muscle protein breakdown. The total muscle supply of glutamine is relatively small (about 240 g), the stress-induced breakdown of 1 kg of muscle tissue provides only 9 g of glutamine, so the catabolic release of glutamine is limited and insufficient with increasing needs. During catabolic stress, a patient with weight 70 kg has a higher intake of glutamine by the intestinal mucosa, kidneys, and immune system than the body can compensate by breaking down its own muscles and increasing glutamine synthesis by about 12 g/day. The real need for glutamine under stress and extreme physical pressure is not less than 18-22 g/day [45].
Hypofunction of skeletal muscles and intolerance to physical activity are distinctive clinical signs of patients with heart failure, coronary heart disease (CHD) and other pathologies of the cardiovascular system (CVS). As a rule, they are more or less associated with a progressive catabolic state, inflammation of skeletal muscles and a violation of oxidative metabolism. For a long period, medical researchers, sports medicine specialists from different countries pay considerable attention to the search for means of optimizing the work of CVS at high physical pressure, with introduction of drugs against coronary heart disease and heart failure, special sports nutrition products. During the search, in particular it was found that polyunsaturated fatty acids (PUFA), such as omega-3, omega-6, omega-9 and others (more than 11) affect the metabolism in the body, including at the cellular level. It turned out that PUFA is not synthesized in the tissues of the body, but necessary for its life. In particular, PUFA protects cells from premature aging and helps to preserve their genetic information. Regulate fat metabolism and vital activity of beneficial bacteria living in the intestine. In 2004, the FDA (US Food and Drug administration) recognized that consumption of some omega-3-PUFA (eicosapentaenoic acid (EPA), docosahexaenoic acid (DHA)) may reduce the risk of coronary heart disease. In the body of peroxide oxidation of polyunsaturated fatty acids counteract the antioxidant system including the glutathione Tripeptide, which is synthesized from glutamine [47-52].
It has been approved that dietary supplements, including fish oil, containing polyunsaturated fatty acids omega-3PNZHK, eicosapentaenoic acid (EPA) idocosahexaenoic acid (DHA), reduce mortality in patients with chronic heart failure [52].
The effect of PUFA was manifested by improvement of cardiac function indices, exercise tolerance and circulating cytokine levels (TNFa, IL-1 and IL-6) in patients with chronic stable heart failure [53].
Moreover, as noted above, G is not only involved in maintaining the homeostasis of nitrogen balance due to changes in the intensity of proteolysis of the muscle, but also in maintaining synthesis of the powerful antioxidant and regulator of cellular integrity of membrane protective, probably warning the rapid oxidation of PUFA [55-58].
In blind, randomized clinical studies in patients with progressive heart failure (HF), which was caused by the progressive development of metabolic disorders, inflammation and atrophy in the myocardium and skeletal muscles, it was noted that there is a clear metabolic transition from the use of normal fatty acids to the use of glucose as the main energy substrate for the generation of ATP in the tricarboxylic acid cycle(CTC). It is assumed that this switching is the result of changes in the genetic program with a decrease in the expression of the enzymes carnitine palmitoyltransferase (CPT) -1, acyl-COA-dehydrogenase with an average chain length (MCAD), citrate synthase, and consequently the use of PUFA [55,58].
This metabolic switching is observed in skeletal muscles with increased (glycolytic) type II muscle fibers and reduced (oxidative) type I fibers [60]. It is assumed that this internal metabolic transition in skeletal muscles contributes to the development of physical exercise’s intolerance.
In the study [55], patients received for long-term (1 and 3 months) ingestion of either a peptide combination and fish oil (source of PUFA) 6.5 g / day, or a combination of safflower oil and milk powder (placebo) in caloric equivalent amounts. Studies have set that the combined Supplement of dipeptide and PUFA did not improve test exercise, muscle properties (biopsy analysis) or muscle function, but increased muscle mass and improved quality of life in patients with chronic stable heart failure. These and other data indicate the potential usefulness of the effects of high doses of PUFA and the effectiveness of simultaneous action of dipeptide supplementation in patients with chronic stable heart failure[61-62]. The question of the mechanisms of interaction and positive influence of dipeptide and polyunsaturated fatty acids on the growth of muscle mass remains: it is not clear whether (actually a dipeptide) Transporter of fatty acids into the cell, whether indirectly by influencing the aggregation of chains of fatty acid molecules with the formation of micelles and those facilitating their absorption in the intestine, or affects the absorption and biosynthesis of other physiologically active substances inside the cells.
Cellular mechanisms of action of PUFA are intensively investigated, but have not yet been determined, but it is believed that omega-3 eicosapentaenoic acid (EPA), docosahexaenoic acid (DHA) can act both directly and through metabolites such as eicosanoids[61,62].
In this regard, there is a reason to consider the mechanisms of modulation of fatty acid oxidation in the intercellular space, within the cell membranes and in the cell cytosol by glutathione as a probable sphere of the control action of AH on the processes of catabolism and absorption of PUFA. The Tripeptide glutathione is a powerful systemic antioxidant, is able to predictably influence the utilization of fatty acids. Although there is currently no scientific evidence of the direct impact of PUFA consumption on the performance and level of physical fitness of athletes, however, some of the identified properties of omega-3 and omega-6 PUFA, indicate the possibility of direct or indirect positive effects of fatty acids on the function of a number of organs and systems during training, accelerate recovery, reduce the short-term or long-term use of the consequences of muscle and ligament injuries, reduce inflammation, stabilize the cardiovascular and respiratory systems at rest and during exercise. Dipeptide favors such action of PUFA [63-66].
In Its turn, glutamine plays an important role in the regulation of glutathione synthesis. It is indicated that glutamine, increasing the formation of glutathione, causes cellular anabolic effects by increasing the volume of cells. On the stress, when the content of free radicals damaging cells is increased in some tissues, the need for glutamine increases [67].
The possibility of direct influence of food intake of enriched AG on increase of glutathione level (GSH) in blood serum and liver tissue was shown experimentally on animals. The relationship between arterial hypertension and the biosynthesis of glutathione (GSH) has been studied as well as the role of communication in protecting the liver. Twenty male Wistar line rats were used, they were randomly divided into two groups: one received standard parenteral nutrition (STD), and the other — with or without hypertension for 7 days. Blood and liver tissue samples were examined after peritoneal injection of hepatotoxin 5-fluorouracil (5-FU). It was found that the concentration of glutathione (GSH b) in serum was significantly higher in the AG group than in the group with STD and the content of GSH in liver tissue was 36.2% higher than in the group with a standard diet, Rats in the AG group had a lower increase in liver enzymes after administration of 5-FU, indicating hepatoprotective effect of dipeptide. I. e. standard food supplemented with AG can protect liver function by increasing the biosynthesis of glutathione and preserving glutathione reserves in liver tissue [68].
Reduced glutathione (GSH) is a Tripeptide composed of the amino acids L-glutamate, L-cysteine, and glycine, it is the most effective antioxidant supporting the intracellular redox potential (or redox potential), which is the ratio of reduced o oxidized (GSSG) forms of glutathione (GSH/GSSG).Maintaining the optimal GSH>>GSSG ratio In a cell is critical for its normal functioning and survival. Glutathione system is a trap of free radicals, in particular, prevents the development of intracellular oxidative stress. GSh protects cells from damage caused by the production of free radicals during physical activity, especially when their levels exceed the body’s ability to defend against them. The lack of GSH puts the cell at risk of oxidative damage[69-71].
Glutathione system of redox potential not only provides resistance of cells to oxidative stress, but also participates in mechanisms of utilization of polyunsaturated fatty acids with subsequent synthesis of cytokines and eicosanoids. Eicosanoids that oxidized derivatives of polyunsaturated fatty acids — eicosatrienoic (C20:3), arachidonic (eicosatetraenoic, C20:4), timnodonic (eicosapentaenoic, C20:5). Eicosanoids are synthesized from omega-3 (ω-3) and omega-6 (ω-6) fatty acids. So, omega-6 are Pro-inflammatory eicosanoid. Edible sources of polyunsaturated fatty acids are vegetable oils, fish oil and omega-3 fatty acids. Eicosanoids are involved in a variety of processes, such as muscle growth, irritation and immune responses to toxins and pathogens. Eicosanoids are formed in almost all cells of the body. But GSH glutathione depletes relatively quickly, especially in conditions of glutamine deficiency, and recovers relatively slowly [72-75].
Especially intensive glutathione is consumed in the kidneys, liver, intestinal mucosa and macrophages. The half-renewal period of GSH (T1/2) in blood plasma is 2 min; in the kidneys 30-50 min, in the liver, intestinal mucosa and macrophages 2-3 h; in the brain; erythrocytes, spleen, lungs 2-4 days; in the lens of the eye 2-8 days [76].
It can be expected that dipeptide not only can affect the absorption and utilization of polyunsaturated fatty acids, synthesis of cytokines and eicosanoids, the synthesis of glutathione, but also as a supplier of glutamine on the synthesis of fatty acids and membrane phospholipids occurring with the participation of metabolites of glutamine, including the substrate of the Krebs cycle acetyl-coenzyme A, which provides acetyl group. Glutamine intake into muscle and liver cells is believed to increase their hydration and serves as an anabolic proliferative signal [77]. Oral administration of dipeptide can change the whole metabolic response of the body to stress.
Dipeptide bioregulator can also be used to correct energy metabolism: both to improve gluconeogenesis in the liver and to Deposit glucose in the form of glycogen. In addition, the intracellular oxidation of glutamine produces ATP, which can be considered as an alternative source of energy at high physical pressure.
The company’s development of an innovative high-quality peptide complex IPH AG and its introduction in recent years in the practice of sports and clinical medicine has expanded the possibilities of prevention and urgent pharmacotherapeutic correction of functions disturbed by overloads, accelerating the processes of rehydration of water and electrolytes, increases the subsequent assimilation of macro- and micronutrients, as well as makes up for the relative deficiency of L-glutamine in extreme physical activity, which during the period of the stressful factor limits or turns off the ability of the intestine to full absorption of organic and mineral substances. Chronic intensive physical activity, without adequate nutrition, metabolic correction and relaxation regimen, leads to a number of gastrointestinal disorders and reduced immunity of fat [78-82]. Due to its exceptional physico-chemical and biochemical properties, IPH AG is indicated for the formation of adequate nutritional support conditions [previous, current and subsequent nutrition] in order to prevent the threat of malnutrition or reduce nutrient uptake, stimulate the increase in lean body weight (LBW) and reduce fat deposition. The high-quality peptide complex produced by the company does not contain impurities, is highly effective and acts in low doses. Thus, IPH AG is able to support a wide range of physiological functions and can become one of the best means of preventing and / or eliminating deficiency conditions. Along with this, being a means of metabolic orientation of action, IPH AG is promising as a remedy of maintaining normal immune function, cellular energy processes in excitable tissues, a protective agent that can counteract the influence of physiological and pathological stress. Forecast, IPH AG will be in demand in sports and clinical medicine, as well as in a wide range of individuals as a practically safe soft adaptoprotective and adaptogenic agent for increasing human resistance to extreme loads.
Literature
А. Dmitriev, А. Kalinichev. SportWiki encyclopedia. sportwikito
- С.N. Lozhkin, А.D. Tikanadze, М.I. Tyuryumina. Bulletin of intensive care. — №4. -2003. — P. 64-69.
- A.A.Lima, G.H.Carvalho, A.A.Carvalho. Nutrition 2002, 18:458-462.
- J. T. Brosnan. The Journal of nutrition. — 2003. — Vol. 133, no. 6 Suppl 1. — P. 2068—2072.
- P.Brosnan, J.Procopio J., M.M.Lima, T.C. Pithon-Curi, R.,Curi. Cell BiochemFunct. 2003;21: 1–9.
- H.Newsholme, F. Abdulkader, E. Rebelato , T. Romanatto, Pinheiro CEJ, R.F. Vitzel. Frontiersin Bioscience, 2011; 16: 315–319.
- J. Antonio ,C. Street . Canadian Journal of Applied Physiology.1999, 24(1): 1-14.
- J.L.Bowtell,K. Gelly , M.L.Jackman . J. Appl. Physiol. 1999, 86(6):1770-1777.
- M. Hakimi , M.A.Mohamadi, Z. Ghaderi. J.Human Sport and Exercise. 2012, 7(4)
- M. Williams . Dietary J. Int. Soc. Sports Nutr. 2005, 2(2): 63–67.
- M. Gleeson . J.Nutr., 2008, 138(10): 2045S-2049
- P. Furst. J.Nutr., 2001, 131(9): 2562S-2568S.
13.YasufumiImamoto, Hisaya Tanaka, Ken Takahashi, Yoshinobu Konno, and Toshiyuki Suzawa. Cytotechnology. 2013 Jan; 65 (1): 135–143.
- Dipeptiven instructions for use of the drug, date of update: 2011.08.23. RDA
- Alanylhistidine (SUSTAIN): the instructions for use. The Internet resource
- P. Klassen, M. Mazariegos , N.W.Mohamadi, P. Furst. The J.of Nutrition (American Society for Nutritional Sciences), 2000, 130:177-182
- A.A.Lima , G.H. Carvalho , A.A. Figueiredo. Nutrition. (2002), 18:458–462.
- E.P. de Oliveira , R.C.Burini, A. Jeukendrup . Sports Med. 2014 May;44 Suppl 1:S79-85.
- P. Furst, K. Pogan, and P. Stehle. Nutrition 1997,13:731-737.
- J. Wernerman, 2004. Clin. Nutr. Suppl. 1:37-42.
21 W.W. Souba . J.P.E.N. 11:569, 1987
- Е.S. Severin, Т.L. Aleynikova, Е.V. Osipov, С.А. Silaeva. Biological chemistry, М. 2008, 364 p.
- E.A.Newsholme, M. Parry-Billings. J. Parenter. Enter. Nutr. 1990;14:63S–67S. doi: 10.1177/014860719001400406.
- J. Wernerman. J. Nutr. 2008;138:2040S–2044S.
- A. Berg , A. Norberg, C.R.Martling,L. Gamrin , O. Rooyackers, J. Wernerman.Intensive Care Med. 2007;33:660–666.
26.B.I. Labow, W.W. Souba, S.F. J. Nutr. 2001;131:2467S–2486S.
- E. Roth . J. Nutr. 2008;138:2025S–2031S.
- J. Tirapegui,V. Cruzat. In: R. Rajendram , V.R.Preedy, V.B. Patel , editors. Glutamine in Clinical Nutrition. Springer; New York, NY, USA: 2015. pp. 499–511.
- V.F. Cruzat ,J. Tirapegui . Nutrition. 2009;25:428–435.
- N.P. Walsh ,A.K. Blannin, P.J. Robson , M. Gleeson . Sports Med. 1998;26:177–191.
- D.G. Rowbottom , D. Keast , A.R. Morton .Sports Med. 1996;21:80–97. doi: 10.2165/00007256-199621020-00002.
32.S. Yoshida ,T.Yunoki , K. Aoyagi, J. Ohta , N.Ishibashi , T.Noake , T. Kakegawa . J. Surg. Res. 59:475, 1995
33.B. Darcy-Vrillon ,L. Posho , M.T. Morel, F. Bernard , F. Blachier F, J.C. Meslin, P.H.Duée . Pediatr. Res. 36:175, 1994
- W.W. Souba, D.W. Wilmorc. Arc. Surg. 120:66, 1985
- La Ding, Li JS. World J Gastroenterol. 2003 Jun;9(6):1327-32.
- W.M. Miner-Williams , B.R. Stevens, P.J. Moughan. Nutr Res Rev. 2014 Dec;27(2):308-29.
- C.R. Harris, J.R. Hoffman, A. Allsopp, N.B.H. Routledge. Nutrition Research, 2012, 1-6
- J.R. Hoffman, H. Stavsky, B. Falk. Int. J. Sports Med. 1995, 16:214-218.
- J.R. Hoffman, N.A. Ratamess, J. Kang, N.Kelly, A.M.Gonzalez, M. Stec, et al. J.Intern.Soc.Sports Nutrition. 2010, 7:8-20.
- J.R. Hoffman, N.A. Ratamess, J. Kang S.L. Rashti.. Kinesiology, 2011, 43(2):125-136.
- K. Brand, Fekl W., J. von Hintenstern, K. Langer, P. Luppa and C. Schoemer. Metabolism 1989. 38:29-33.
- Z. Spolarics , C. H. Lang, G. J. Bagby , J. J. Spitzer. Am. J. Physiol. 261:G185, 1991
- K. Brand. Biochem. J. 228:353, 1985
- K. Brand, W. Leibol, P. Luppa , C. Schoerner, A. Schulz Immunobiology 1986.173:23
- https://himija-online.ru/wp-content/ — интернет ресурс
- N. Piccirillo , S. De Matteis , L. Laurenti , P. Chiusolo , F. Sorà, M.Pittiruti , S.Rutella, S. Cicconi , S.Sica . Haematologica. 2003 Feb;88(2):192-200.
- B.Levine , J. Kalman , L. Mayer , H.M. Fillit , M.P. Packer . The New England journal of medicine. 1990;323:236–241. [PubMed] [Google Scholar]
- A.Chokshi , K. Drosatos , F.H. Cheema , Ji R, T.Khawaja , Yu S, T. Kato , R. Khan , H.Takayama ,R. Knoll , H.Milting , C.S.Chung , U. Jorde , Y. Naka , D.M. Mancini, I.J.Goldberg , P.C. Schulze. Circulation. 2012;125:2844–2853.
- W. Doehner , M. Rauchhaus , P. Ponikowski , I.F. Godsland , von Haehling S, D.O.Okonko , F. Leyva , A.J.Proudler , A.J. Coats, S.D.Anker . J. Am Coll Cardiol. 2005;46:1019–1026.
5O. H. Drexler , U. Riede , T. Munzel , H. Konig , T.Funke, H. Just. Circulation. 1992;85:1751–1759.
- R.Hambrecht, P.C. Schulze, S. Gielen , A. Linke , S. Mobius-Winkler S, Yu J, J.J. Kratzsch , G. Baldauf , M.W. Busse, A. Schubert , V. Adams , G. Schuler. J.AmCollCardiol. 2002;39:1175–1181
- H.F Gissi., L.Tavazzi L, A.P. Maggioni, R. Marchioli, S. Barlera, M.G.Franzosi . Lancet. 2008;372:1223–1230.
- S.Nodari , M. Triggiani , U. Campia , A. Manerba, G.Milesi , B.M. Cesana. J Am Coll Cardiol. 2011;57:870–879.
- G. Smith, P.Atherton, D.N. Reeds, B.S. Mohammed, D Rankin., M.J. Rennie, B. Mittendorfer. The American journal of clinical nutrition. 2011;93:402–412.
- K.Shahzad , A. Chokshi , P.C. Schulze. Current clinical pharmacology. 2011;6:288–294.
- J.M., Engel, J.Muhling , M.Kwapisz , M.Heidt. Acta anaesthesiologica Scandinavica. 2009;53:1317–1323.
- V. Carubelli , A. Castrini , V. Lazzarini , M.Gheorghiade , M. Metra, C.Lombardi . Heart Fail Rev. 2015;20:39–51.
- G.S. Francis , B.H. Greenberg, D.T. Hsu, B.E. Jaski, M. Jessup , M.M. LeWinter , F.D.Pagani , I.L. Pina, M.J. Semigran, M.N. Walsh, D.H. Wiener, C.W. Yancy. Circulation. 2010;122:644–672.
- B. Levine, J.Kalman , L.Mayer, H.M. Fillit, M.P. Packer. The New England journal of medicine. 1990;323:236–241.
- B.S. Christina Wu, T. S. Kato, Ji. Ruiping, Cynthia Zizola.
Circ Heart Fail. 2015 Nov; 8(6): 1077–1087.
- Jump DB, Depner CM, Tripathy S. Omega-3 fatty acid supplementation and cardiovascular disease. JLipidRes. 2012;53:2525–2545.
- R.T.Holman. J Nutr. (1998)128 (2): 427S–433S.
- “Editorial: Are PUFA harmful?”. Br Med J. 4 (5883): 1–2. 1973. PMID 4755208.
- M. E. Jørgensen , P. Bjeregaard , K.Borch-Johnsen . Diabetes care. — 2002. — Vol. 25, no. 10. — P. 1766—1771
- Christina Wu, Tomoko S Kato, Ruiping Ji, Cynthia Zizola, Danielle L Brunjes, Yue Deng, Hirokazu Akashi, Hilary F Armstrong, Peter J Kennel, Tiffany Thomas, Daniel E Forman, Jennifer Hall, Aalap Chokshi, Matthew N Bartels, Donna Mancini, David Seres, P Christian Schulze. Journal Scan / Research · August 31, 2015
- R.W. Hong, J.D. Rounds,W. S. Helton, M. K. Robinson. Ann. Surg. 215:114, 1992
- Jyh-CherngYu, Z.M. Jiang, D.M. Li. World Journal of Gastroenterology 5(2) · April 1999 , 33
- V.I. Kulinskiy, L.S. Kolesnuchenko . Biomedical chemistry, 2009 vol 55, edition. 3, p. 255-277.
- C.H. Lillig, C. Berndt. (2013) Glutaredoxins in thiol/disulfide ex-change, Antioxidants & Redox Signaling, 18, 1654–65.
- V.V. Ivanov, Е.V. Shakhristova, Е.А. Stepovaya, О.L. Nosareva, Т.S. Fedorova, N.V. Ryazantseva, V.V.Novitskiy. Biochemistry, 2015, vol 80, edition. 1, p. 111 – 122.
- E.S. Severin, edition Biochemistry: Maual for medical schools. Moscow: GEOTAR-MED, 2003.
- H. Gerster. Can adults adequately convert alpha-linolenic acid (18:3n-3) to eicosapentaenoic acid (20:5n-3) and docosahexaenoic acid (22:6n-3)?. Int J Vitam Nutr Res. 1998 68 (3): 159–73.
- J.T. Brenna. CurrOpin Clin NutrMetab Care 2002, 5 (2): 127-32. PMID 11844977.
- R.T. Holman . JNutr. 1998, 128 (2): 427S-433S. PMID 9478042.
- V.I. Kulinskiy, L.S. Kolisnichenko. Advances in biological chemistry, 1990, 31, p. 157-179.
- P. Furst, K. Pogan , P. Stehle P. Nutrition 1997; 13:731-737
- A. V. Dmitriev, L. M. Gunina. Fundamentals of sports nutrition. SPb., 2018, pp. 194-204
- Roth E. immune and cellular modulation by amino acids. Klin. The bellies. 2007, 26: 535-544.
- Roth E. non-nutritional effects of glutamine. The journal of nutrition. 7th workshop on the evaluation of amino acids. 2008, 138: 2025-2031.




